

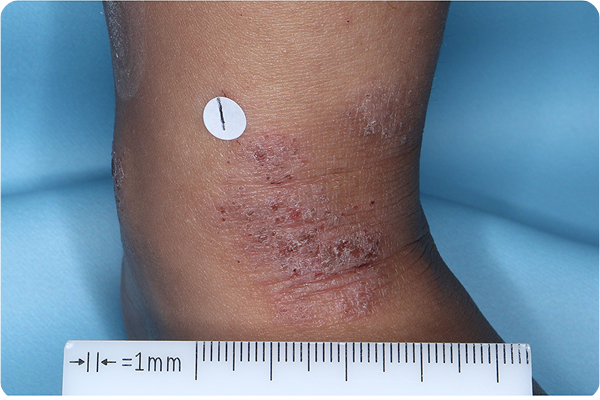
Primary Endpoint Success†7
Age: 3
Gender: Male
Fitzpatrick Skin Type: V
Patient: 1032-009

Primary Endpoint Success†7
Age: 5
Gender: Male
Fitzpatrick Skin Type: V
Patient: 1028-021
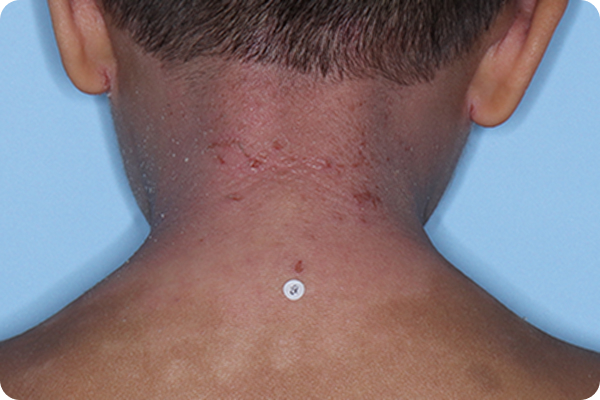
Primary Endpoint Success†7
Age: 3
Gender: Male
Fitzpatrick Skin Type: IV
Patient: 1028-009
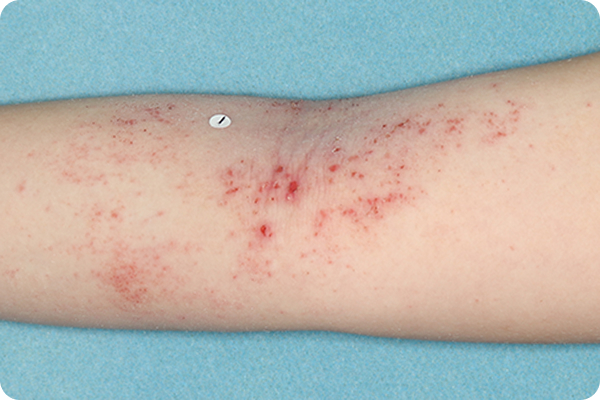
Primary Endpoint Success†7
Age: 9
Gender: Female
Fitzpatrick Skin Type: IV
Patient: 1020-002
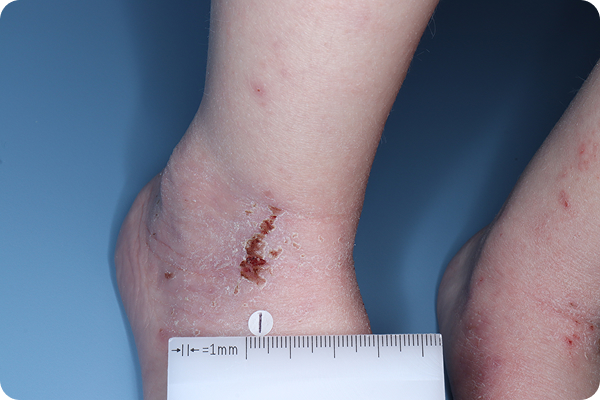
Primary Endpoint Success†7
Age: 3
Gender: Male
Fitzpatrick Skin Type: II
Patient: 1028-007
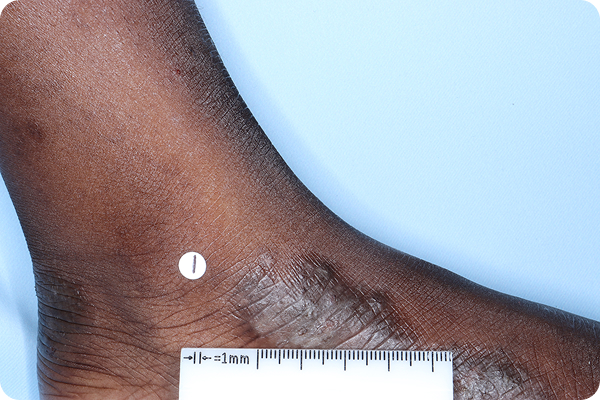
Primary Endpoint Success†7
Age: 7
Gender: Male
Fitzpatrick Skin Type: VI
Patient: 2016-013
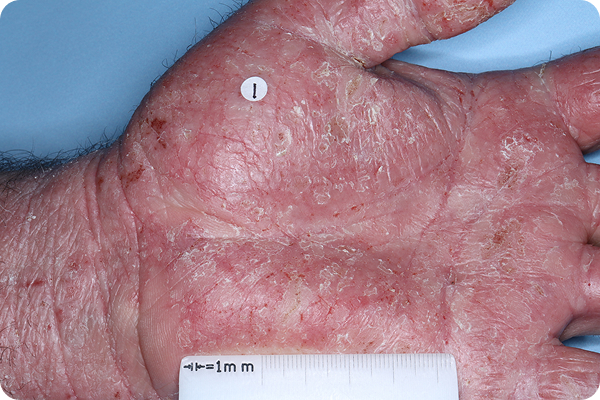
Primary Endpoint Failure‡7†7
Age: 54
Gender: Male
Fitzpatrick Skin Type: II
Patient: 1905-027

Primary Endpoint Failure‡7†7
Age: 4
Gender: Female
Fitzpatrick Skin Type: II
Patient: 2005-007
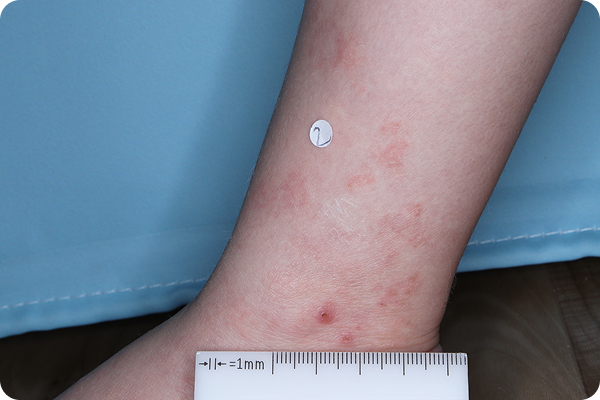
Primary Endpoint Failure‡7†7
Age: 3
Gender: Female
Fitzpatrick Skin Type: I
Patient: 1032-007
*Please note that these photographs are representative of one target lesion on individual patients treated with VTAMA cream during the pivotal clinical studies.
†Primary endpoint success defined as vIGA-AD score of 0 (clear) or 1 (almost clear) and ≥2-grade improvement from baseline at Week 8.2
‡Primary endpoint failure defined as failure to achieve vIGA-AD score of 0 or 1 (clear or almost clear) and ≥2-grade improvement from baseline at Week 8.2
Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD): measure of AD severity with scores ranging from 0 (clear) to 4 (severe). vIGA-AD is a trademark of Eli Lilly and Co.
Eczema Area and Severity Index (EASI): measure of disease severity that incorporates body surface and lesion intensity into a composite score from 0 to 72, with higher scores indicating more severe disease.
Patient Oriented Eczema Measure (POEM): 7-item questionnaire to measure eczema severity focusing on the disease as experienced by the patient. Scores range from 0 to 28, with higher scores indicating more severe disease.
Peak Pruritus-Numeric Rating Scale (PP-NRS): patients are asked to rate their worst itch in the past 24 hours on a scale of 0 (no itch) to 10 (worst itch imaginable).
*Please note that these photographs are representative of one target lesion on individual patients treated with VTAMA cream during the pivotal clinical studies.
†Primary endpoint success defined as vIGA-AD score of 0 (clear) or 1 (almost clear) and ≥2-grade improvement from baseline at Week 8.2
†Primary endpoint failure defined as failure to achieve vIGA-AD score of 0 or 1 (clear or almost clear) and ≥2-grade improvement from baseline at Week 8.2

INDICATION: VTAMA® (tapinarof) cream, 1% is an aryl hydrocarbon receptor (AhR) agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older.
SELECTED SAFETY INFORMATION
Adverse Events: In atopic dermatitis, the most common adverse reactions (incidence ≥1%) were: upper respiratory tract infection, folliculitis, lower respiratory tract infection, headache, asthma, vomiting, ear infection, pain in extremity, and abdominal pain.
INDICATION: VTAMA® (tapinarof) cream, 1% is an aryl hydrocarbon receptor (AhR) agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older.
SELECTED SAFETY INFORMATION
Adverse Events: In atopic dermatitis, the most common adverse reactions (incidence ≥1%) were: upper respiratory tract infection, folliculitis, lower respiratory tract infection, headache, asthma, vomiting, ear infection, pain in extremity, and abdominal pain.
Before prescribing VTAMA cream, please read the Prescribing Information.
