


VTAMA cream is the only aryl hydrocarbon receptor (AhR) agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older
See the Science
VTAMA is the first and only AhR agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older.
Across all disease severities, skin tones, and treatment areas. In the 8-week pivotal studies, up to 46% of patients achieved clear or almost clear skin compared to 18% on vehicle.1,2,*
Clinically proven to deliver sustained results, even during treatment-free months.
*In ADORING 1 and ADORING 2, vIGA-AD success, defined as vIGA-AD=0 or 1 and ≥2-grade improvement from baseline at Week 8, was achieved by 45% and 46% of patients on VTAMA vs 14% (P<0.0001) and 18% (P<0.0001) of patients on vehicle, respectively.1
†Average duration of first treatment-free interval. Intent-to-treat population, observed case.3
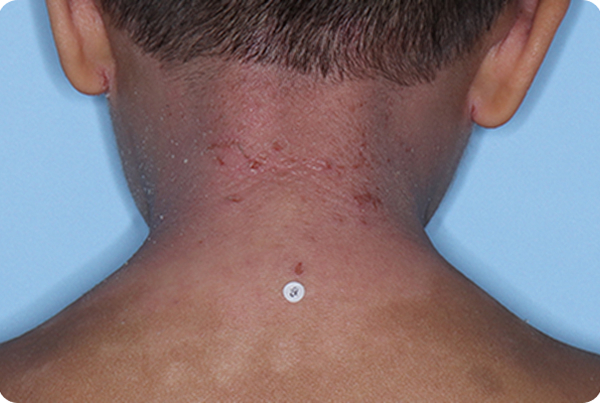
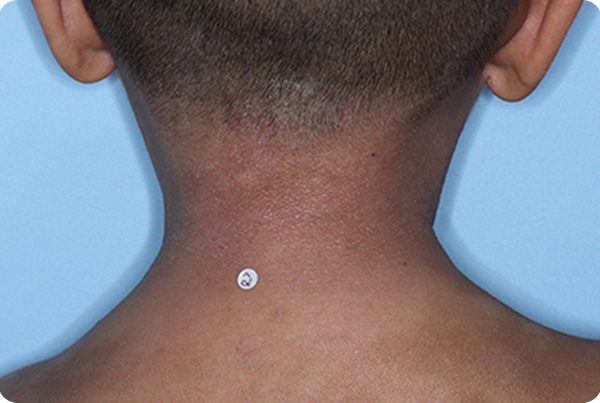
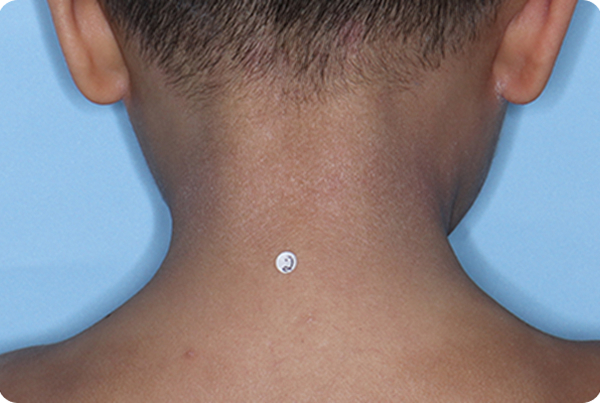
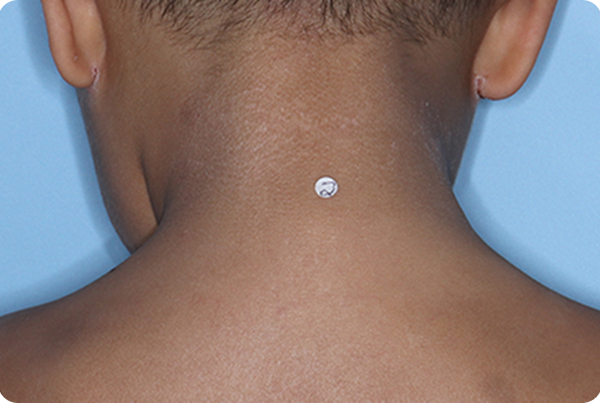

INDICATION: VTAMA® (tapinarof) cream, 1% is an aryl hydrocarbon receptor (AhR) agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older.
SELECTED SAFETY INFORMATION
Adverse Events: In atopic dermatitis, the most common adverse reactions (incidence ≥1%) were: upper respiratory tract infection, folliculitis, lower respiratory tract infection, headache, asthma, vomiting, ear infection, pain in extremity, and abdominal pain.
INDICATION: VTAMA® (tapinarof) cream, 1% is an aryl hydrocarbon receptor (AhR) agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older.
SELECTED SAFETY INFORMATION
Adverse Events: In atopic dermatitis, the most common adverse reactions (incidence ≥1%) were: upper respiratory tract infection, folliculitis, lower respiratory tract infection, headache, asthma, vomiting, ear infection, pain in extremity, and abdominal pain.
Before prescribing VTAMA cream, please read the Prescribing Information.
