

VTAMA cream provided skin clearance and disease control of atopic dermatitis for patients down to 2 years of age.1

728 patients with mild, moderate, or severe atopic dermatitis entered the ADORING 3 48-week, open-label, long-term extension (LTE) study of VTAMA.
Patients who entered the study with vIGA-ADTM≥1 used VTAMA daily and patients with vIGA-AD=0 stopped VTAMA. If patients achieved vIGA-AD=0 during the study, they stopped VTAMA until vIGA-AD≥2 occurred. When vIGA-AD≥2 occurred, patients restarted VTAMA.1
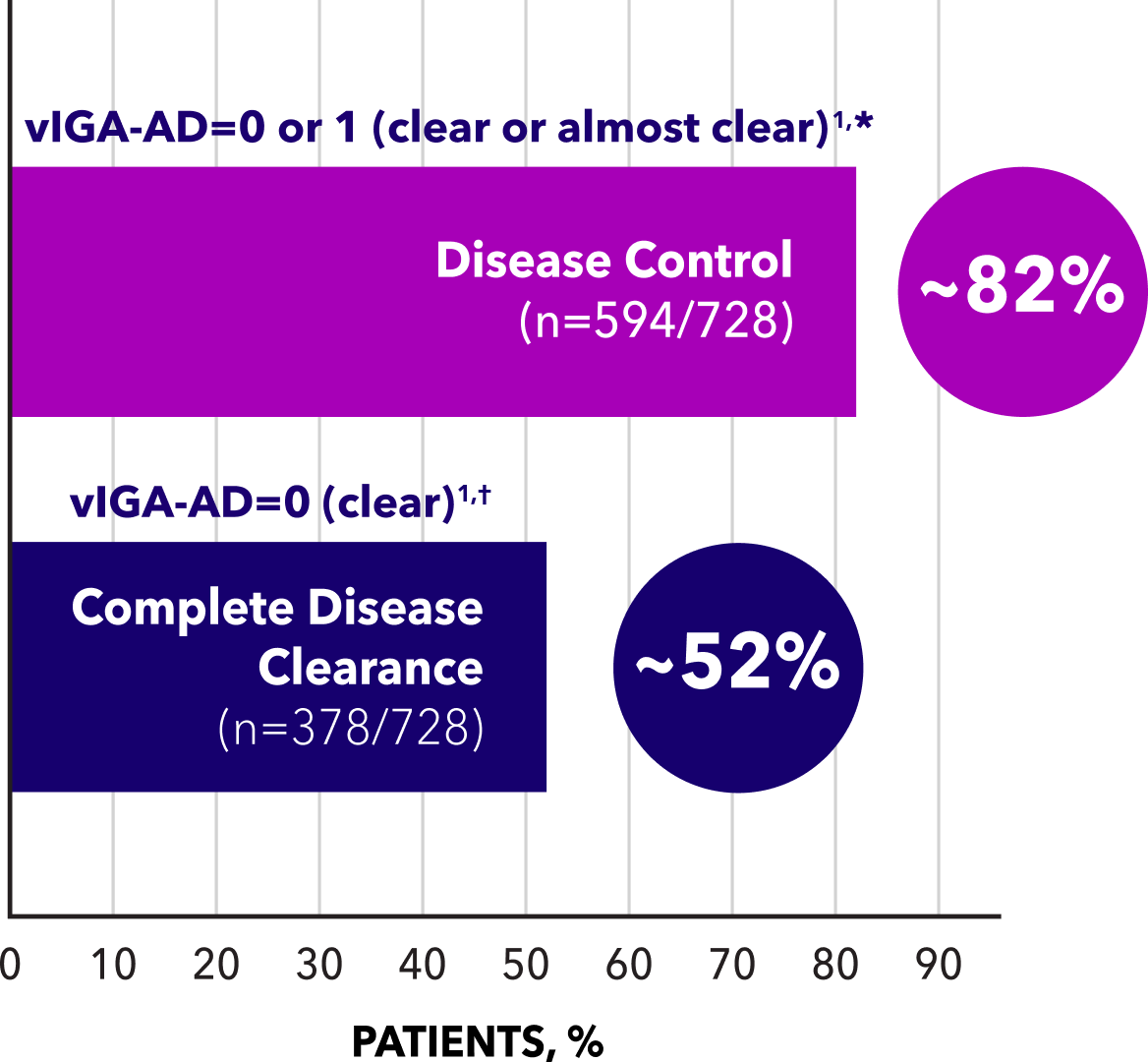
In the 48-week, open-label, LTE study, atopic dermatitis patients (n=378) who entered with or achieved clear skin (vIGA-AD=0) maintained an average duration of ~80 consecutive days during the first treatment-free interval1,‡
*Including patients who entered with vIGA-AD=0 (n=58) or vIGA-AD=1 (n=189), and vIGA-AD≥2 who achieved 0 or 1 (n=347).1
†Including patients who entered with vIGA-AD=0 (n=58) and patients entering with vIGA-AD≥1 who achieved vIGA-AD=0 at least once during the open-label LTE study (n=320).1
‡Maintenance of clear or almost clear skin (vIGA-AD=0 or 1) off treatment, after first achieving complete disease clearance (vIGA-AD=0) and discontinuing treatment.
There were no new safety signals with long-term tapinarof use in the LTE study, including in young children1
TEAE, treatment-emergent adverse event.
§Includes folliculitis, follicular eczema, keratosis pilaris, milia, and application site folliculitis.1

INDICATION: VTAMA® (tapinarof) cream, 1% is an aryl hydrocarbon receptor (AhR) agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older.
SELECTED SAFETY INFORMATION
Adverse Events: In atopic dermatitis, the most common adverse reactions (incidence ≥1%) were: upper respiratory tract infection, folliculitis, lower respiratory tract infection, headache, asthma, vomiting, ear infection, pain in extremity, and abdominal pain.
INDICATION: VTAMA® (tapinarof) cream, 1% is an aryl hydrocarbon receptor (AhR) agonist indicated for the topical treatment of atopic dermatitis in adults and pediatric patients 2 years of age and older.
SELECTED SAFETY INFORMATION
Adverse Events: In atopic dermatitis, the most common adverse reactions (incidence ≥1%) were: upper respiratory tract infection, folliculitis, lower respiratory tract infection, headache, asthma, vomiting, ear infection, pain in extremity, and abdominal pain.
Before prescribing VTAMA cream, please read the Prescribing Information.
